New Drug May Be Better Psoriasis Treatment
 By Dennis Thompson HealthDay Reporter
By Dennis Thompson HealthDay Reporter
MONDAY, April 26, 2021
A breakthrough psoriasis drug is better at dealing with the itchy and unpleasant skin disease than medications by now on the industry, in accordance to outcomes from two scientific trials.
There was a “evening and day change” in the outcomes from bimekizumab in comparison against two founded psoriasis medicines, secukinumab (Cosentyx) and adalimumab (Humira), reported Dr. Mark Lebwohl, a co-researcher in one particular of the scientific trials.
“We’ve by no means had a drug which in its section 3 trials had additional than fifty% of sufferers attain” a one hundred% reduction in their psoriasis indicators, reported Lebwohl, dean for scientific therapeutics at the Icahn University of Medicine at Mount Sinai in New York Town.
“We are now at a point wherever we can obvious the vast the greater part of psoriasis sufferers with medicines that are pretty efficient and pretty risk-free,” he added.
Based mostly on these outcomes, Lebwohl expects the Belgian pharmaceutical firm UCB Pharma to pursue speedy approval of bimekizumab with the U.S. Food items and Drug Administration.
“I would hope it would be on the industry this summer season,” he reported.
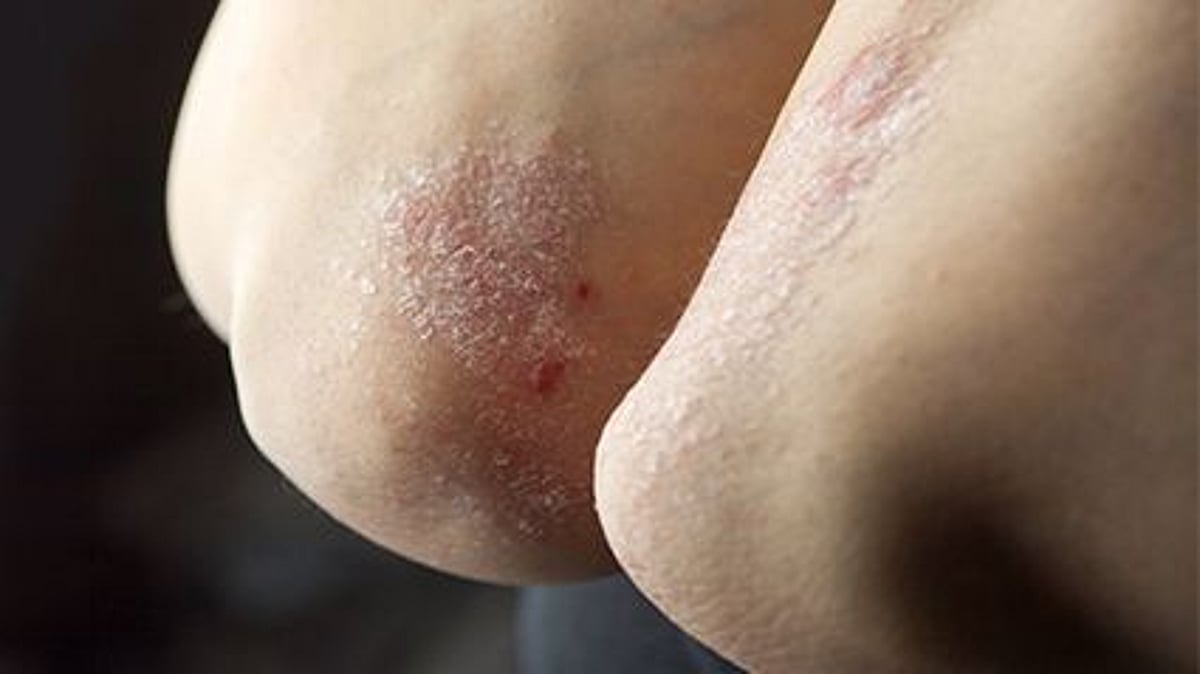
Psoriasis impacts additional than eight million folks in the United States, in accordance to the Nationwide Psoriasis Foundation.
It’s an autoimmune disease that speeds up skin mobile advancement, causing cells to pile up on the floor of the skin and type plaques that itch, melt away and sting. These plaques can seem on any aspect of the overall body, but are most generally found on the elbows, knees and scalp.
A professional-inflammatory biochemical known as interleukin-17 (IL-17) has been implicated in the advancement of psoriasis, Lebwohl reported. Secukinumab and adalimumab operate by blocking the chemical’s most potent type, known as IL-17A.
Bimekizumab blocks both IL-17A and another type of the chemical known as IL-17F, Lebwohl reported. The injectable drug is administered once a thirty day period.
“The biology [of the two sorts of IL-17] is overlapping — 17A is additional potent but 17F is additional considerable,” Lebwohl reported. “Even though 17A is more robust at causing psoriasis, there is additional of 17F. By blocking both, you get the entire impact.”
Following forty eight months of treatment method, about 67% of bimekizumab sufferers had comprehensive clearing of their psoriasis plaques, in comparison with 46% of sufferers receiving secukinumab, in accordance to outcomes of the trial that Lebwohl co-authored. A complete 743 sufferers participated.
The other scientific trial, involving 478 sufferers, offered very similar outcomes. Following 16 months, 86% of sufferers on bimekizumab had experienced a 90% reduction in their psoriasis plaques, virtually double the 47% who accomplished the exact same response with adalimumab.
“They block IL-17A, when this blocks both IL-17A and IL-17F,” Lebwohl reported. “That is in all probability why it can be so efficient. Blocking that further minor bit of IL-17 really receives you the added performance.”
Bimekizumab also has been demonstrated to effectively deal with psoriatic arthritis, a problem that impacts one in 3 folks with psoriasis, Lebwohl reported.
Persons taking bimekizumab were four to 10 occasions additional likely to have a reduction in their arthritis indicators than a placebo group, with the response developing with the measurement of the dose, in accordance to outcomes revealed in The Lancet.
Blocking IL-17 does lead to a better risk of yeast infections, and the risk is more robust with bimekizumab than the other two medicines, outcomes confirmed.
“Mother nature has done an experiment for us by giving us folks who are deficient in IL-17, and they get dreadful yeast infections,” Lebwohl reported. “We expected prior to the analyze is that the only aspect impact we might see was yeast infections, and that is what happened.”
The delicate to average situations of yeast an infection that transpired in the scientific trials were “very easily treated with fluconazole,” an oral anti-fungal drug, Lebwohl reported.
SLIDESHOW
Forms of Psoriasis: Medical Photographs and Treatment options
See Slideshow
Dr. Michele Environmentally friendly, a skin doctor with Lenox Hill Healthcare facility in New York Town, reviewed the findings.
“This is an extraordinary analyze demonstrating considerable outcomes utilizing an interleukin-17 inhibitor to deal with plaque psoriasis,” she reported.
On the other hand, Environmentally friendly sounded a notice of caution, urging further analyze of the drug.
“A greater sample measurement needs to be made use of due to the fact in addition to candidiasis, interleukin inhibitors have been connected with better fees of other opportunistic infections, intense infections and cancer,” Environmentally friendly reported.
The scientific trial outcomes were revealed April 23 in the New England Journal of Medicine, and also were offered at an on-line assembly of the American Academy of Dermatology.
UCB Pharma funded both trials.
A lot more data
The Nationwide Psoriasis Foundation has additional about psoriasis.
Sources: Mark Lebwohl, MD, dean for scientific therapeutics, Icahn University of Medicine at Mount Sinai, New York Town Michele Environmentally friendly, MD, skin doctor, Lenox Hill Healthcare facility, New York Town New England Journal of Medicine, April 23, 2021

Copyright © 2021 HealthDay. All rights reserved.


