Trending Clinical Topic: Remdesivir
What your medical doctor is reading through on Medscape.com:
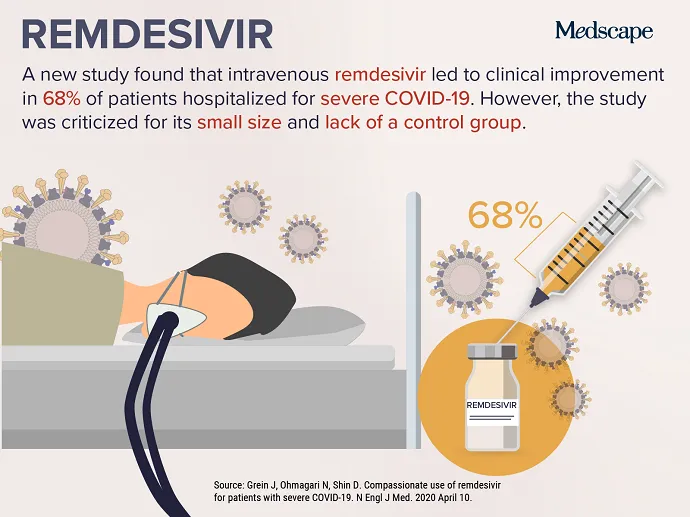
APRIL 24, 2020 — As the COVID-19 pandemic carries on, attempts to identify a risk-free and helpful therapy have intensified, leading to this week’s top trending clinical topic. New facts on the investigational antiviral drug remdesivir were published April 10 in the New England Journal of Medication . A lot more than two thirds of severely ill patients with COVID-19 who had been supplied remdesivir for “compassionate use” improved immediately after receiving the medicine. Of the fifty three patients bundled in the review, 22 had been in the United States, 22 had been in Europe or Canada, and nine had been in Japan. Patients acquired a 10-working day study course of remdesivir, consisting of two hundred mg administered intravenously on working day one, adopted by one hundred mg everyday for the remaining nine days of treatment method.
Despite the fact that some had been inspired by the benefits, many others warned that limitations in the review make deciphering the facts complicated. Josh Farkas, MD, an intensivist from Vermont, provided 11 causes why the review “reveals nothing” and termed the paper “hot garbage” on Twitter. Other doctors lamented the erosion of the peer-evaluation course of action through the COVID-19 pandemic. The absence of a management team and a small client sample dimension have been frequently cited as major issues.
In a letter to Gilead, the enterprise at the rear of remdesivir, John Mandrola, MD, calls for blinding and a placebo arm to be added to foreseeable future trials. Despite the fact that he applauds the attempts produced consequently far, Mandrola reiterates that these additions would make the findings a lot more responsible, as “they would be absolutely free from the probable biases of clinicians—who, out of the motivation to have an out there treatment method, might consciously or unconsciously make or delay conclusions to lower oxygen and stay clear of mechanical air flow in patients in the remdesivir arm.”
At this position, an skilled panel of the Infectious Health conditions Society of The us (IDSA) states evidence is insufficient to propose any probable pharmacologic therapies for regimen use in patients with COVID-19. The exertion to validate a treatment method tactic is huge and international. The Globe Wellness Business has identified a record of “promising candidates” for COVID-19 treatment method, such as remdesivir, lopinavir-ritonavir, immunotherapies, and convalescent sera. Around 60 trials involving these therapies have been prepared, are recruiting, or have by now started. As the benefits of all those trials turn into out there, the drugs analyzed will receive enormous interest, just as remdesivir did this week.
‘)
} else
// If we match both of those our exam Topic Ids and Buisness Ref we want to location the advert in the center of web page one
if($.inArray(window.s_topic, moveAdTopicIds) > -one && $.inArray(window.s_small business_reference, moveAdBuisRef) > -one)
// The logic down below reads count all nodes in web page one. Exclude the footer,ol,ul and table factors. Use the varible
// moveAdAfter to know which node to location the Advert container immediately after.
window.placeAd = purpose(pn)
var nodeTags = [‘p’, ‘h3′,’aside’, ‘ul’],
nodes,
goal
nodes = $(‘.write-up-web page:nth-child(‘ + pn + ‘)’).locate(nodeTags.join()).not(‘p:empty’).not(‘footer *’).not(‘ol *, ul *, table *’)
//goal = nodes.eq(Math.ground(nodes.size / 2))
goal = nodes.eq(moveAdAfter)
$(”).insertAfter(goal)
// Now passing in one to transfer the Advert in to web page one
window.placeAd(one)
else
// This is the default place on the base of web page one
$(‘.write-up-web page:nth-child(one)’).append(”)
})()
$(purpose()
// Make a new conatiner the place we will make our lazy load Advert connect with if the access the footer segment of the write-up
$(‘.major-container-3’).prepend(”)
)
